What are examples of metals, non-metals and metalloids?
4.7 (717) · € 15.00 · En Stock
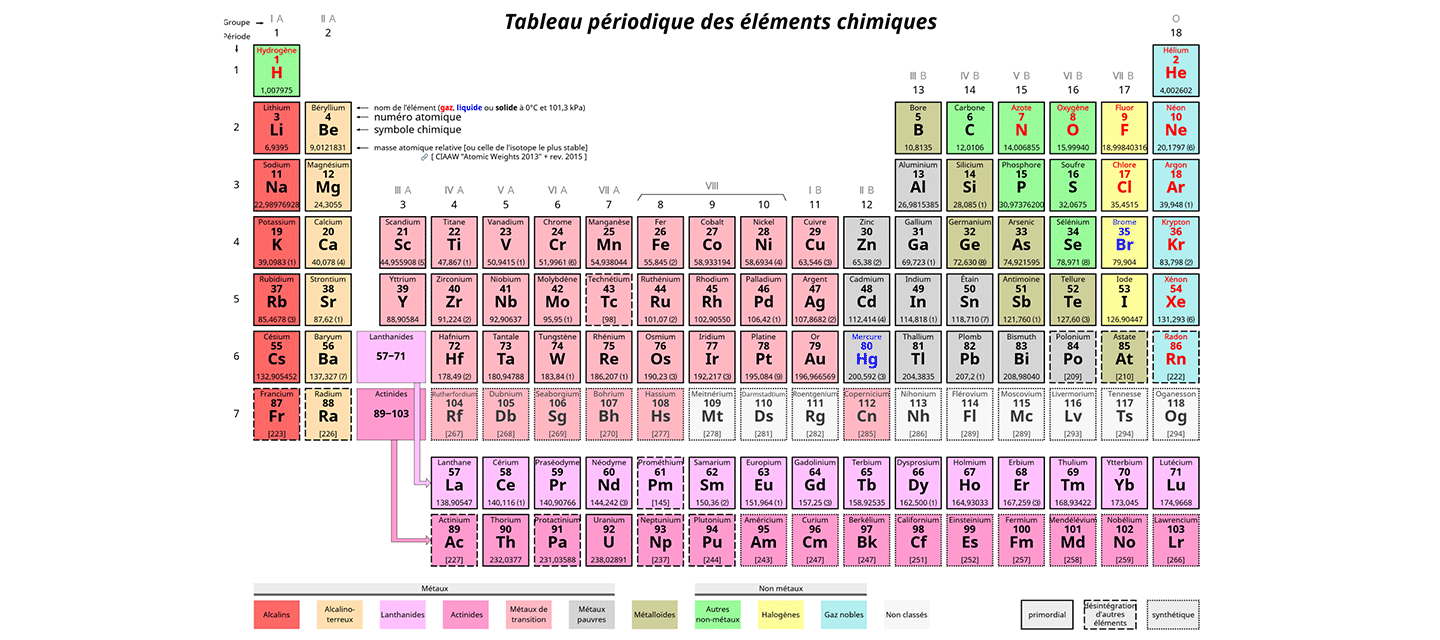
Metals like sodium are found on the left side of the periodic table. Non metals like Oxygen are found on the left side of the periodic table. Metalloids like Silicon are found between the metals and nonmetals. Metals lose electrons to become positive ions. The elemental metals bond using metallic bonding which uses a sea of electrons to bond unlimited numbers of atoms together. Most elements are metals. magnesium, iron, silver are examples. Metals have low electronegativity and want to lose electrons. Non metals gain electrons to become negative ions. The elemental nonmetals bond using covalent bonds. There are fewer nonmetals that metals. The noble gases, Oxygen, Nitrogen are examples. Nonmetals have high electronegativity and want to gain electrons. Metalloids can go either direction becoming either positive or negative. There are very few metalloids. examples are Carbon, Silicon.

Lesson Video: Metals, Nonmetals, and Metalloids

Metals, Metalloids, and Nonmetals - Element Classification Groups

Metals, Non Metals and Metalloids - Meaning & Difference - Teachoo

Periodic Table Metals and Non-Metals
What are some interesting facts about metals and non metals? - Quora

Metals, non metals, and metalloids

Metals, Nonmetals, and Metalloids Crossword - WordMint

Metals, Nonmetals and Metalloids activity
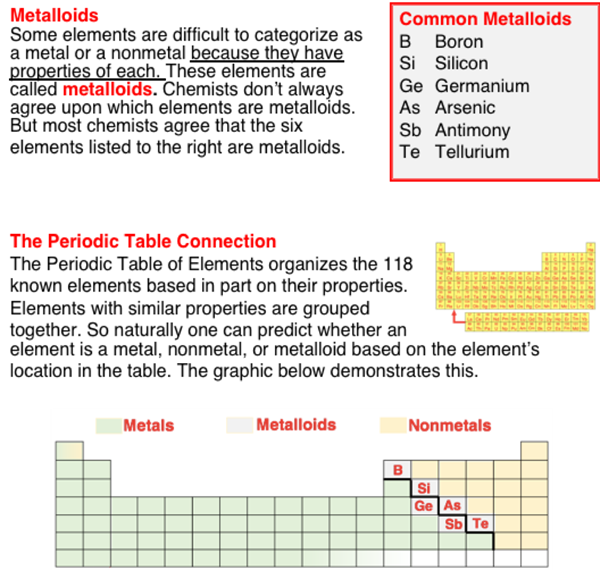
Metals, Nonmetals, and Metalloids - Help

What are examples of metals, non-metals and metalloids?
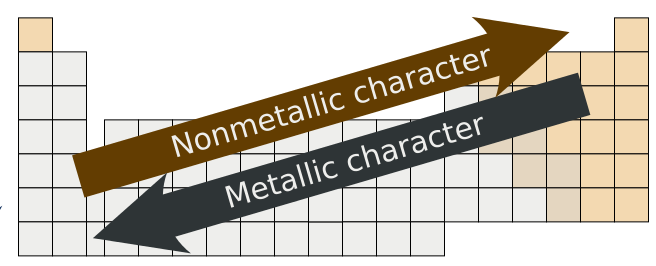
7.6: Metals, Nonmetals, and Metalloids - Chemistry LibreTexts