What Is the Density of Air at STP?
4.9 (433) · € 15.50 · En Stock
This is a look at the density of air at STP, the factors that affect air density, and how it is changed by temperature, pressure, and water vapor.

Density of a Gas at STP Lesson # ppt download

CHEM 105 Problem Set 22 - Problem Set 22 – Mixtures of Gases Chem 105 (a) What is meant by standard - Studocu

Answered: calculate the r value based on density…
Air is a mixture of N_2 and O_2 having 20
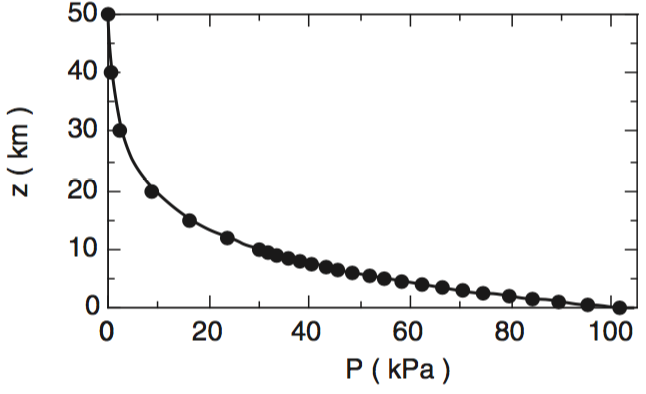
UBC ATSC 113 - Standard Atmosphere-Pressure

Use the molar volume of a gas at STP to calculate the density (in

Answered: 4. Calculate the R value based on…

Air Properties Definitions

50. The density of air is 0.00130 g/mL. The vapour density of air will be (a) 0.00065 (b) 0.65 (c) 14.4816 (d) 14.56

SOLVED: What is the density of He at STP? Why do helium-filled balloons rise in air?

PPT - Unit V: The Mole Concept PowerPoint Presentation, free download - ID:5896038

The density of air is 0.001293 g/cc at STP. Its vapour density is - NEETLab

Question Video: Calculating the Volume of Ammonia Gas at STP Given the Mass
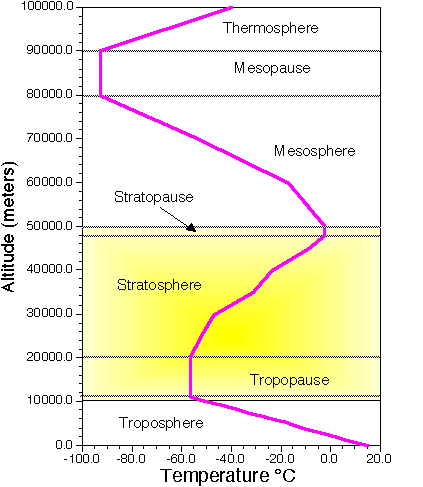
Atmo336 - Fall 2012

Real gases






:max_bytes(150000):strip_icc()/nature-3294543_1920-50b0ec12ca0b4385b87bcd7723b3d97d.jpg)





