Particulate Matter Inspection - Pharmaceutical & Medical
4.6 (734) · € 34.50 · En Stock
Identify unwanted particulate matter in glass vials and ampoules to prevent contaminated products from reaching consumers with Cognex Deep Learning.

Particulate Matter Testing of Cardiovascular Devices

Ophthalmic Product Package Inspection Using Vacuum Decay Technology
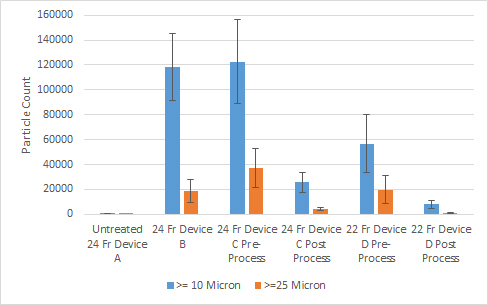
Frontiers Cardiovascular medical device evaluation of particulate following simulated use testing

The need for new control strategies for particulate matter in parenterals: Pharmaceutical Development and Technology: Vol 24, No 6

Visual Inspection of injectable products: why the visible particulate matter test is required, blog

Case Study: Particle Detection Challenges in Pharmaceutical Cleanroom

GMP Testing Laboratories & Services - Pace Analytical

Vial and Ampoule Body Inspection - Pharmaceutical & Medical

Reworking Sterile Drug Products - BioProcess InternationalBioProcess International

Particulate Contamination in Single-Use Systems: Challenges of Detection, Measurement, and Continuous Improvement - BioProcess InternationalBioProcess International

Difficult-to-Inspect Drugs Require New Processes
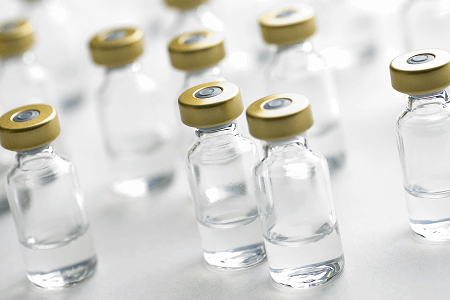
Achieving Zero Particulates In Parenteral Manufacturing What Needs To Be Done

Coating Testing and Particulate Testing

Particulate Matter Testing – MED Institute