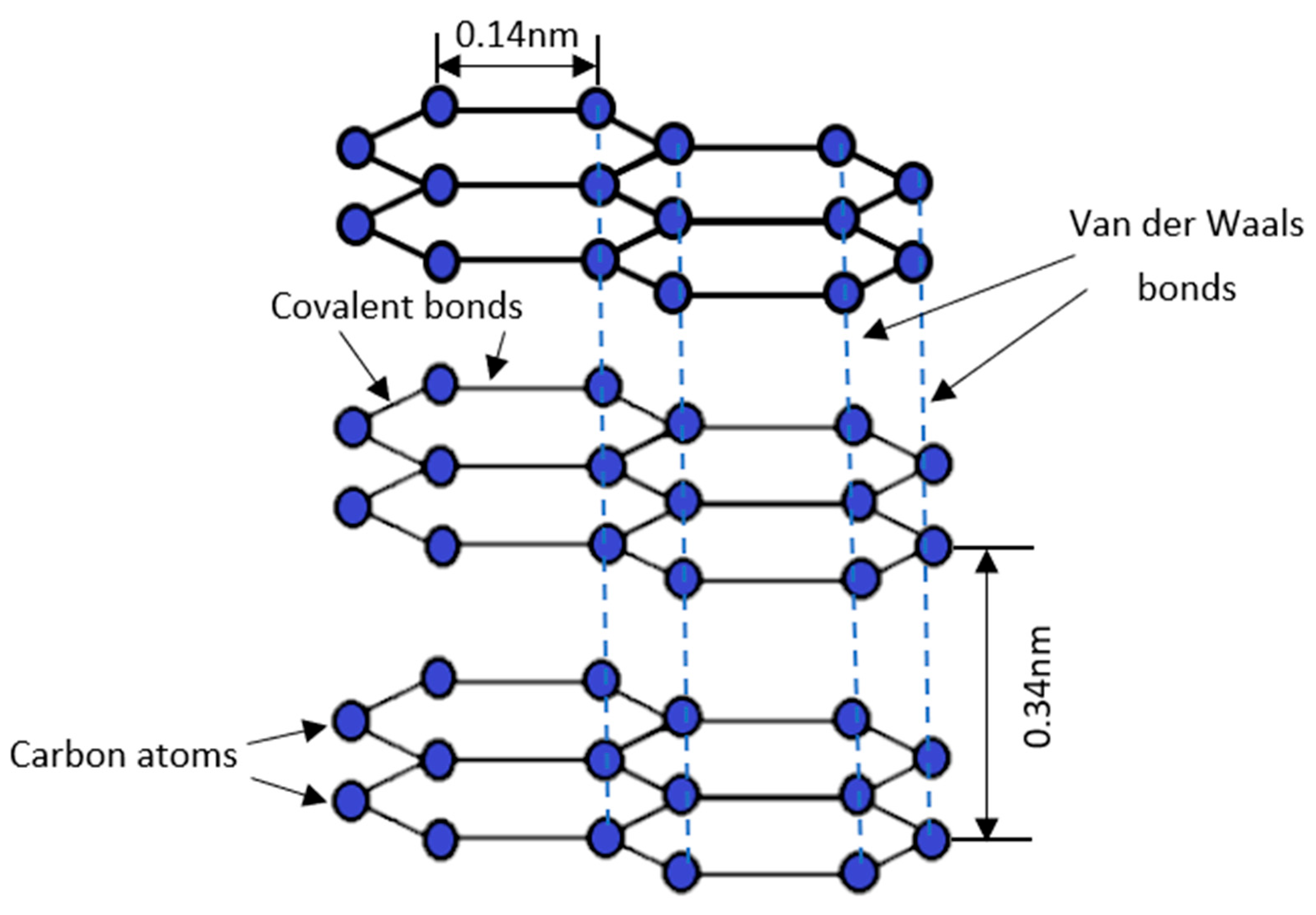
Does The Difference In Structure Make Graphite Soft But Diamond Hard?
5 (453) · € 25.99 · En Stock

Weird Form of Carbon Acts as Reversible Diamond—A First
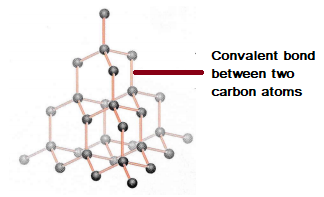
Structure and Bonding of Carbon - Chemistry Revision

Why is diamond hard while graphite is soft?
Why is graphite soft and diamond hard? - Quora

Diamond Description
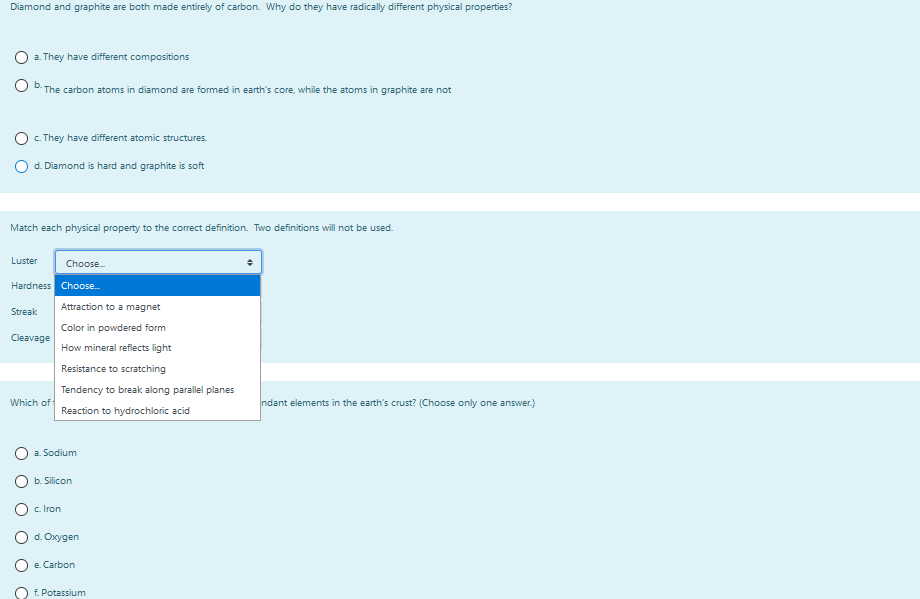
Solved Diamond and graphite are both made entirely of carbon

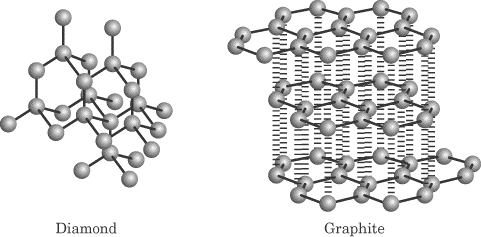
Treasures of the Earth, Molecular Structures of Diamond and Graphite

Structural comparison of carbon allotrophs- carbon & its compounds, ICSE chemistry Std-8
Properties of solids
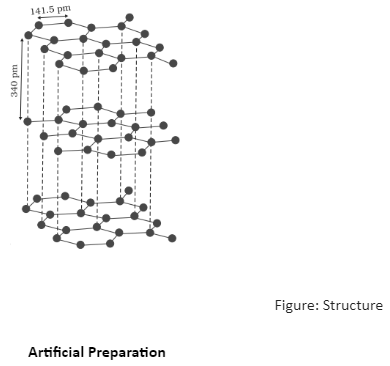
Graphite

Why is diamond the hardest common substance while graphite is a soft, solid lubricant?
Tu pourrais aussi aimer
© 2018-2024, thefforest.co.uk, Inc. ou ses affiliés